FDA
During an ECRI-hosted webinar on AI-enabled diagnostics, a panel of experts discussed the promise and risks that come with the technology for both physicians and patients alike.
Small Biz Bill A Big Win For Medtech Innovation: Bipartisan Legislation Awaits President’s Signature
The US Congress overwhelmingly passed legislation reauthorizing federal programs essential to smaller medtech firms in need of funding. AdvaMed says the bill is critical for medtech innovation.
The US FDA has issued an early alert concerning Intuitive Surgical staplers used during various procedures with the da Vinci Surgical System. The company is instructing customers to stop using the devices.
A recent FDA report to Congress reflects ongoing safety issues with orthopedic and cardiac implants, as well as commodities such as syringes and PPE.
The Trump EPA wants to loosen restrictions the Biden administration placed on facilities that sterilize medical devices with ethylene oxide, arguing the rollbacks are necessary to ensure device accessibility and supply chain security. Opponents to the rollbacks say EtO is a danger to public health.
During the latest round of MDUFA negotiations, the FDA and medtech industry agreed to include language in the next commitment letter that would provide direction for the use of carryover funds from user fees. The parties also found agreement on several other topics.
As AI becomes ubiquitous in diagnosing diseases, clinicians must balance the technology’s enormous upside, such as improving the speed and accuracy of diagnoses, with the potential for errors that can lead to delayed or missed treatment.
The FDA’s device center has received a second citizen petition relating to the NarxCare algorithm by Bamboo Health. Both petitions argue that NarxCare qualifies as a regulated clinical decision support product, but it is not clear whether the second petition will succeed where the first failed.
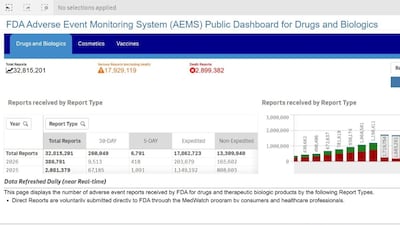
The US FDA is replacing its individual product type adverse event reporting portals with a unified database, known as AEMS. While adverse event reports for some sectors such as drugs and vaccines joined the new portal on March 11, devices, human foods and tobacco products will be added in May.
HeartBeam’s CEO says the strength of clinical data convinced the US FDA to reverse course and grant clearance to his company’s innovative ECG device, which he says can assess arrhythmias at home with hospital accuracy.
Recently published lists show that the US Food and Drug Administration had cleared almost 2,000 digital health tools by the end of 2025. Of these, well over half – 1,104 – were radiology devices incorporating AI. Find a full breakdown in these tables.
During MDUFA negotiations held Feb. 11, the medtech industry agreed to a proposal from the FDA to base its hiring commitments on MDUFA IV.
The US FDA is warning that anti-choking devices can prove ineffective in emergencies and that methods established by the American Red Cross and the American Heart Association to remove blockages from choking victims should be tried first.
The US FDA has issued an early alert concerning Abiomed’s Impella purge cassettes, a critical heart pump component that delivers rinsing fluid to the Impella catheter.
The latest round of MDUFA VI negotiations discussed different perspectives between the FDA and industry on TAP 2.0 and the agency’s proposals for enhancing the program.
In two warning letters posted Feb. 24, the US FDA cited a device manufacturer with clinical trial violations and another for failing to meet current good manufacturing practices.
The US Supreme Court struck a blow to the Trump administration’s efforts to impose sweeping tariffs, ruling the president exceeded his authority. Limiting tariffs has been a priority for the medtech industry.
The US FDA has a new digital health head in radiologist Rick Abramson, an AI-industry veteran who is now director of the Digital Health Center of Excellence. Abramson has been an FDA adviser since last summer.
Class I medical device recalls reached record-breaking levels in 2025, according Sedwick’s annual product safety report, with device failure the leading cause.
A new report from the Senate HELP Committee offers regulatory and legislative reforms aimed at offering patients faster access to medical innovations, including changes to AI and digital health regulation.