Asia Pacific
A decade ago, China was buying Western drugs. Now it is selling them. Executives who ignore the shift do so at their peril.
Formycon bagged a third commercialization opportunity for its pembrolizumab candidate FYB206, with Lotus Pharmaceutical taking over rights for the APAC region.
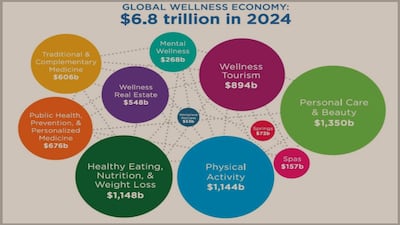
“Wellness real estate has been the leading contributor to wellness economy growth globally, as well as among many high-growth countries that also fall into the top 25 largest markets,” GWI reports.
Pilatus Biosciences' Switzerland-Taiwan-US structure exemplifies how biotechs might navigate US-China tensions, leveraging Chinese manufacturing efficiency while maintaining American investor credibility and partnership opportunities.
The disposal of the Asia-Pacific business outside China to BGH Capital allows Aspen to refocus on core growth drivers, including GLP-1 products and restoring profitability in sterile manufacturing.
New Zealand is also lifting its prohibition on advertising unapproved medicines at medical conferences and trade shows in order to boost its appeal as a host for such events and generate millions of dollars in revenue.
The top awards at the annual MedTech Innovator APAC are handed out to an increasingly varied range of products and applications, and the class of 2025 was no exception in its reflection of healthtech’s ever-widening innovator base.
The culmination of the MedTech Innovator APAC awards program 2025 comes later this month when the top prizes are handed out at the grand final in Singapore. But all cohort companies benefit from the competition’s mentoring process, says awards organizer Fredrik Nyberg.
Novartis’s APMA chief outlines the company’s tailored approach to deliver long-term outcomes, novel access solutions via innovative financing options and shape the CVD ecosystem in the extraordinarily diverse region. Partnering to build AI infrastructure is another key prong.
Neuraxpharm has launched its Australian affiliate as part of its continued international expansion beyond Europe. The company will introduce CNS treatments including its acquired Nuvigil, Provigil, and the recently TGA-approved Briumvi for multiple sclerosis.
Novotech's CEO talks about rising interest for trials in regions with “regulatory agility” and “strategic insulation” from geopolitical complexity, especially among emerging and mid-sized biotech sponsors. Operational "reassessment” among some sponsors on exposure to China-based CROs/CRDMOs is another area he discussed.
In the final part of an interview with Scrip, Menarini’s Asia-Pacific CEO talks about the group’s digital capabilities, including their impact on a shortage issue in Australia, and leveraging RWE to inform healthcare practices across the region. The Italian company is also revving up its business development and alliance engine.