Belarus
Country
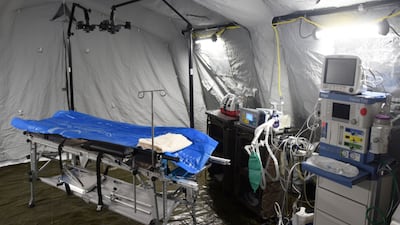
Ukraine’s medical supply needs have evolved since Russia launched its military invasion of the country on 24 February. EU healthcare decision makers and the medical technology industry have since stood firm in their support of the besieged country, said MedTech Europe’s Jesús Rueda.
Ukraine’s medical supply needs have evolved since Russia launched its military invasion of the country on 24 February. EU healthcare decision makers and the medical technology industry have stood firm in their support of the besieged country, said MedTech Europe’s Jesús Rueda.
Russia and Ukraine had been key players in the pharma markets of Central Asian and surrounding countries before the launch of the Russian invasion into its neighbor on 24 February. Because of this, both countries are poised to give ground in this growing regional market.
Pharmaceutical leaders are responding to the many and varied challenges arising from the war in Ukraine.
Given the ongoing conflict in Ukraine, EU medtech notified bodies have been told to comply with sanctions intended to hurt companies based in Russia and Belarus.
After a five-year regulatory transition, access to the Eurasian Economic Union market for new medtech products must now take place via the EAEU system only. But having got off to a slow start, further system transitional measures are now under discussion.
The Eurasian Union’s medical device regulatory system, many years in the planning, becomes mandatory in January 2022. But new amendments posted in late summer allow for national registrations to remain valid after the deadline. Moscow-based consultancy RegMT explains the background, and the likely path ahead.
The Medicines Patent Pool has announced that the first shipments of dolutegravir under a fresh agreement covering upper-middle-income countries have reached Azerbaijan and Belarus.
There are just 4.5 months left until the new harmonized Eurasian Economic Union medtech system becomes mandatory, signifying major changes in regional market access. EAEU member Russia is meanwhile strengthening its post-market medtech controls.
The Medicines Patent Pool is giving interested parties until 18 December to submit license applications for the HIV treatment dolutegravir in four middle-income countries, following announcement of the agreement last month.
Stada will pay $660m for a basket of 20 brands from Takeda to expand its consumer health portfolio in Russia and a number of strategically important CIS markets.
The new regulatory framework in the Eurasian Economic Union is now in place and the first drug approval applications have been filed under the mutual recognition system. Applications are expected to rise significantly in 2019 once IT systems are fully in place and more experience has been gained.