Canada
Country
AHPA Awards Hero, Champion, Visionary and others at annual meeting; Kimberly-Clark’s information chief has major-merger experience; RäFoods’ marketing chief has history with CEO; and Eshbal adds VP for North America expansion.
Sandoz says GLP-1s represent a long-term growth opportunity but warns the off-patent market will take time to develop, with early launches potentially coming in Canada and Brazil as the company tests supply dynamics and regulatory timelines.
B+L also plans by March 1 to start distributing its Blink dry eye drop line extension, Triple Care preservative free, and in the first half of the year to file an NDA in US for a Luxe formulation of its Lumify redness eye drop brand.
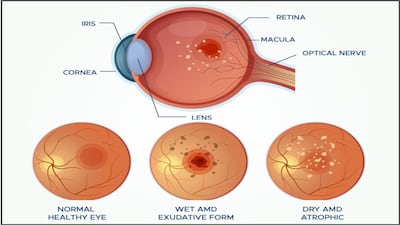
Sandoz’s aflibercept biosimilar Enzeevu launch in Canada comes just a few days after the firm signed a multiple asset deal, including an undisclosed ophthalmology product, with Alvotech, which recently gained immediate launch for its Eylea rival in the country.
Sandoz and Alvotech have struck a deal that will see the firms collaborate on multiple biosimilar candidates for the Australian, Canadian and New Zealand markets across therapeutic areas including ophthalmology, immunology and gastroenterology.
Alvotech has ticked off all of the remaining patent-related matters for its aflibercept biosimilar, with multiple market entries expected in 2026.
Dr Reddy’s gears for semaglutide debut across markets, including India and Canada, where Novo Nordisk has second brands of the GLP-1 agonist potentially ready. Can the Indian group close out compliance queries from the Canadian regulator soon?
Generics Bulletin reviews global regulatory developments across the world.
Dr Reddy’s gears for semaglutide debut across markets, including India and Canada, where Novo Nordisk has second brands of the GLP-1 agonist potentially ready. Can the Indian group close out compliance queries from the Canadian regulator soon?
Generics Bulletin reviews global regulatory developments across the world.
Formycon has again stepped closer to financial certainty with another pembrolizumab biosimilar deal. This time, it unlocked a significant opportunity for the US and Canadian markets.
Korean firm’s Eydenzelt biosimilar win full label alignment with Eylea, including all approved ophthalmic indications.