Consumer
“In a very siloed organization made up of a jigsaw of different companies, no one was thinking enterprise first,” says Perrigo CEO Patrick Lockwood-Taylor. “The fundamentals of running operations, brand-building, innovation, they are now as they were.”
At opening session of CHPA’s annual Self-Care Leadership Summit on March 17 in Orlando, it presented Fuad Sawaya its highest honor named for Ivan Combe, an industry pioneer who served on its board from 1958 until 2000.
As Stada awaits the imminent closure of a deal that will see it come under the ownership of CapVest, the firm has reported 2025 sales that grew by 6% to €4.3bn, boosted by a double-digit biosimilars increase that drove Specialty sales above €1bn for the first time.
Antonio Martinez Descalzo has more than 20 years' experience in life sciences, health and nutraceutical industries, including with food and beverage giant ADM. Israeli firm has completed pilot production and is moving into full manufacturing.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
US Trade Representative is taking two swings under Section 301(b) of the 1974 Trade Act. Customs and Border Protection has March 19 to provide US Court of International Trade with next update on its progress on providing refunds for IEEPA tariffs.
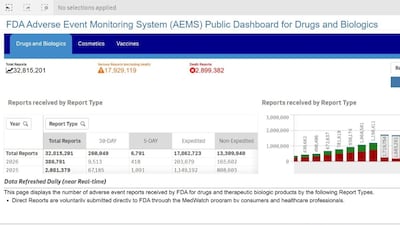
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
FTC’s sending checks totaling more than $40,700 to 578 consumers who purchased deceptively marketed treatment plans from Golden Sunrise between July 2017 and July 2020. Company owner and chief medical officer both pleaded guilty to criminal charges.
US Court of International Trade expects by March 12 to receive from Customs and Border Protection a “short report describing the progress” it “has made toward the development of a process to issue refunds of IEEPA duties paid with interest.”
Delaying or altogether eliminating limits on all THCs in lawful hemp Congress approved in 2025 could come down to either House Appropriations AG/FDA subcommittee, chaired by a proponent for lower limits, or Energy and Commerce Health subcommittee, chaired by a supporter for eliminating lower limits.
VALR Energy names appoints chief revenue officer; CEO at Audien Hearing; Anderson retiring at NACDS; Lindora appoints president and chief medical officer; and USHR expands leadership.
Industry groups acknowledge a March 27 public meeting has potential to influence how firms can expand product lines with additional ingredients and formulations allowed under DSHEA. FDA acknowledges industry’s interest in introducing additional ingredients.