Cosmetics
Bart Heldreth, Cosmetic Ingredient Review executive director, reflects on the accomplishments of its expert safety panel, which has published safety assessments for more than 7,000 ingredients since CIR’s establishment in 1976.
At opening session of CHPA’s annual Self-Care Leadership Summit on March 17 in Orlando, it presented Fuad Sawaya its highest honor named for Ivan Combe, an industry pioneer who served on its board from 1958 until 2000.
Campaign launched with video featuring Jang Won-Young away from her glamorous stage image in quiet, everyday moments to reflect the brand's philosophy, "When Skin Heals, You Bloom."
US Trade Representative is taking two swings under Section 301(b) of the 1974 Trade Act. Customs and Border Protection has March 19 to provide US Court of International Trade with next update on its progress on providing refunds for IEEPA tariffs.
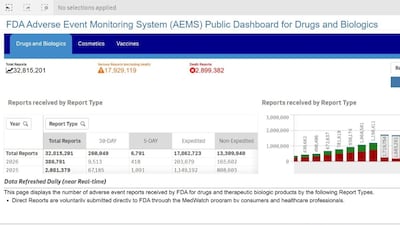
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
US Court of International Trade expects by March 12 to receive from Customs and Border Protection a “short report describing the progress” it “has made toward the development of a process to issue refunds of IEEPA duties paid with interest.”
“The president is certainly hamstrung in many ways by this ruling. For businesses, there's still just sort of a lot of general uncertainty in regard to what else is the president going to try to do, what other authorities might he try to pull,” says Tax Foundation analyst Alex Durante.
Kenvue’s full-year net sales 2.1% dip to $6.38bn came largely on a 2.2% organic sales decline driven by a 2.3% volume decrease but slightly offset by 0.2% foreign currency benefit.
User fee program changes traditionally are technical and FDA-specific, but the Trump Administration appears to also want policies advocating its “America First” agenda included.
“Your firm’s quality systems are inadequate” or “these products are unapproved new drugs” each firm warned with reference to FDA’s regulation for GMPs for pharmaceuticals, supplements or cosmetics. Warnings included 10 to firms about GMP violations topical drugs production.
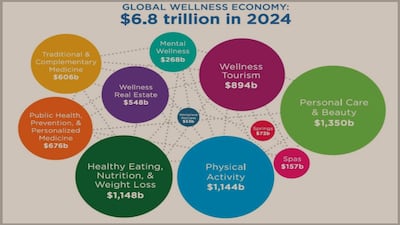
“Wellness real estate has been the leading contributor to wellness economy growth globally, as well as among many high-growth countries that also fall into the top 25 largest markets,” GWI reports.
In contributed commentary, Susan Levy, SBL Consulting Group founder and principal, discusses likely health and wellness market developments which would reflect a steady shift toward more accessible, consumer-centric models of care complementing traditional healthcare.