Drug Approval Standards
“New Clinical Investigation Exclusivity (3-Year Exclusivity) for Drug Products: Questions and Answers” draft guidance discusses statutory and regulatory criteria for three-year exclusivity eligibility and provides recommendations on content and format of requests.
FDA final rule effective date is March 7, 2033, when it will assign new 12-digit NDCs and convert all previously assigned 10-digit NDCs for OTC and Rx drugs to the 12-digit NDC format and start a three-year clock for compliance through March 6, 2036.
A new regulatory reliance procedure in New Zealand is expected to better allow companies to include the country in their global drug launch plans more easily.
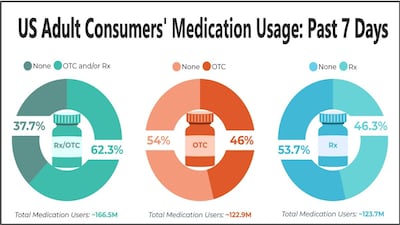
“Observed changes in use prevalence align with changes in regulatory status and availability, including medications” available Rx-only during a 2002 study, “available OTC during the current study period.”
“Our sense is that companies are currently focused on timing, scope and execution. For example, knowing when a final order might issue … We also imagine that formulators are scrutinizing the proposed list of permitted combinations, the 6% cap, and the permitted dosage forms,” say ArentFox Schiff att
Agency says FY 2026 Tier 1 OMOR fees are $587,529 and Tier 2 fees are $117,505. The fees, due to FDA when an OMOR is filed, aren’t included in OMUFA target revenue calculation for each fiscal year based entirely on annual facility registration fees.
CDER Office of Generic Drugs publishes MaPP for prescription-to-nonprescription switches and ANDAs to explain regulatory responsibilities for makers of generic copies of reference listed drugs approved for OTC switch.
Sunscreen products industry and public health advocacy groups have been critical that FDA has not approved a new filter since 1999 even as countries in Europe and other regions allow using numerous additional ingredients in sunscreens.
FDA’s proposed order follows its review of OMOR DSM-Firmenich submitted showing bemotrizinol, at concentrations up to 6%, is generally recognized as safe and effective and can be added as an active ingredient to sunscreen monograph.
CDER Office of Generic Drugs publishes MaPP for prescription-to-nonprescription switches and ANDAs to explain regulatory responsibilities for makers of generic copies of reference listed drugs approved for OTC switch.
FDA says Teresa Michele, in CDER nonprescription drugs program leadership roles since 2013, was being moved to another position in the agency.
In announcement of approving VKT Pharma’s application, FDA included storage and handling instructions common in labeling, suggesting concerns persist about the potential for NDMA to form after products are distributed.