E-Commerce
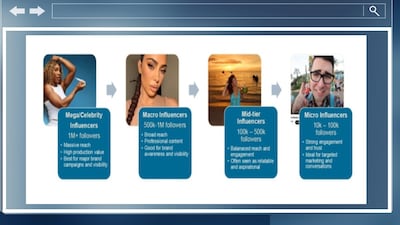
Attorneys in charge of advertising for healthcare and beauty product firms join IZEA Worldwide CEO in discussing impact influencers can have for brands and responsibilities marketers have for content they share during NAD conference.
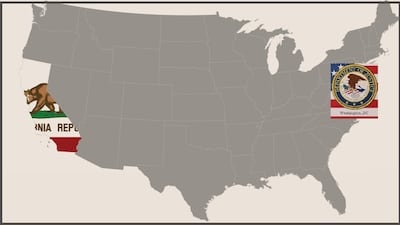
Trade group tells DoJ about complaint against New York law age-restricting sales of supplements containing ingredients labeled or promoted for weight loss and bodybuilding. Compliant manufacturers adhere to federal safety standards for heavy-metal content, but California regulation could incentivize
Mark Meador marks fifth month as FTC member quoting Bible, lamenting agency’s loss of a key consumer protection tool and referencing US jobs lost when businesses move them overseas in his remarks at advertising regulation conference in Washington.
Three FDA warnings related to supplement manufacturing, labeling or marketing submitted to Amazon or companies selling vitamin, mineral or supplement products on its e-commerce platform since April 2024.
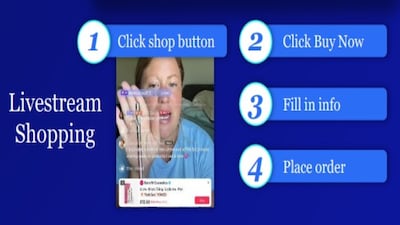
“It is shifting so quickly. It's shifting underneath our feet,” says NielsenIQ wellness VP Sherry Frey. Even comparing the past year to the preceding eight, “it's just accelerating quicker and quicker, and one of the biggest areas is online.”
Direct seller has deals to fully acquire and 51% ownership of Link BioSciences, which formulates personalized supplements. It also announced a deal to acquire Pruvit Ventures, ketone supplements direct seller.
AI prowess “will not be a competitive advantage long-term. It's going to be table stakes. But there are people who are going to get there sooner and they will have an advantage for a while,” says C&D CEO Matthew Farrell. P&G’s investment in AI, meanwhile, produced its “Great Idea Generator proprietary platform.”
After launching Retaine MGD Advanced, OcuSoft says a release by Bruder Healthcare referenced Retaine MGD trademark and statements from a previous OcuSoft announcement about the original product attributed to an optometrist. B+L, Rohto brand and homeopathic firm Relief Products also make US OTC eye care space moves.
Perelel acquiring Loom online platform providing reproductive health, sexual wellness and parenting education. First direct-to-consumer product for Curive Healthcare’s Plum brand formulated for vaginal area dryness.
Streamlined process for reporting problems is key piece of “unified Human Foods Program” which officially launched on 1 October, as Commissioner Robert Califf says, “a new model for field operations and other modernization efforts.”
Liquid herbal blends firm WishGarden Herbs expands its leadership with three hires; ChromaDex appoints Ozan Pamir CFO; Vivazen adds CMO; Post Malone promotes poppi; and marketing consultant Gould gets ‘Most Influential’ nod.
Council for Responsible Nutrition contends New Jersey bill, which Assembly Health Committee amended with substantial language to make restrictions more stringent on 23 September, is targeted as inaccurately as restrictions in New York effective in April.