FDA
Bart Heldreth, Cosmetic Ingredient Review executive director, reflects on the accomplishments of its expert safety panel, which has published safety assessments for more than 7,000 ingredients since CIR’s establishment in 1976.
Serial patent litigations have become a loophole in the Hatch-Waxman Act, disbalancing the scale of innovation and competition, said Jon Potter, executive director of the newly established Coalition Against Pharma Patent Abuse, in an exclusive Generics Bulletin interview.
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
Alvotech is shifting toward dual-source manufacturing and operational execution as it addresses FDA setbacks, expands its biosimilar pipeline and drives growth through global launches, with US approvals representing key upside to its 2026 outlook.
NRx says the FDA has identified no bioequivalence deficiencies in its Ketafree ANDA, clearing a key hurdle ahead of a potential mid-2026 decision as the firm pursues parallel generic and innovative ketamine strategies.
A series of surprise rejections has made for an FDA that drug and vaccine manufacturers are finding much harder to read than in years past.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
The US FDA released one of the highest numbers of product-specific guidances for generic development in a single batch.
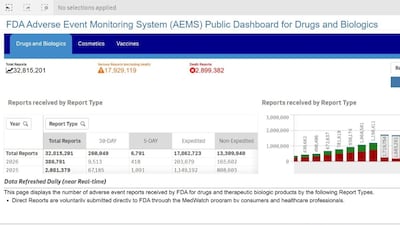
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
The latest biosimilars guidance from the FDA – which promises to further streamline applications by making it easier for developers to rely on non-US comparator products – has been warmly received by the off-patent industry.
Delaying or altogether eliminating limits on all THCs in lawful hemp Congress approved in 2025 could come down to either House Appropriations AG/FDA subcommittee, chaired by a proponent for lower limits, or Energy and Commerce Health subcommittee, chaired by a supporter for eliminating lower limits.
The FDA offered to relax the onshoring criteria to receive a priority ANDA review in GDUFA IV, but could drop another America First proposal.