OTC Drugs
“Four or five major acquisitions over the last few years repositioned it, a 140-year-old company, from being a B-to-B company to B-to B-to-C company. That is a major enterprise strategy shift,” says CEO Patrick Lockwood-Taylor.
“In a very siloed organization made up of a jigsaw of different companies, no one was thinking enterprise first,” says Perrigo CEO Patrick Lockwood-Taylor. “The fundamentals of running operations, brand-building, innovation, they are now as they were.”
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
At opening session of CHPA’s annual Self-Care Leadership Summit on March 17 in Orlando, it presented Fuad Sawaya its highest honor named for Ivan Combe, an industry pioneer who served on its board from 1958 until 2000.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
US Trade Representative is taking two swings under Section 301(b) of the 1974 Trade Act. Customs and Border Protection has March 19 to provide US Court of International Trade with next update on its progress on providing refunds for IEEPA tariffs.
AHPA Awards Hero, Champion, Visionary and others at annual meeting; Kimberly-Clark’s information chief has major-merger experience; RäFoods’ marketing chief has history with CEO; and Eshbal adds VP for North America expansion.
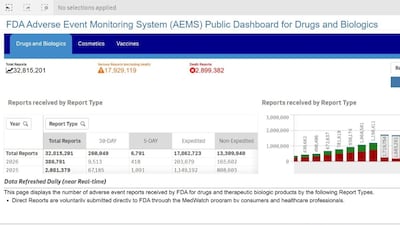
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
US Court of International Trade expects by March 12 to receive from Customs and Border Protection a “short report describing the progress” it “has made toward the development of a process to issue refunds of IEEPA duties paid with interest.”
The agency offered to commission a third-party study of efficiency, workload and other factors to determine whether the base PDUFA revenue should be lowered mid-program cycle.
“New Clinical Investigation Exclusivity (3-Year Exclusivity) for Drug Products: Questions and Answers” draft guidance discusses statutory and regulatory criteria for three-year exclusivity eligibility and provides recommendations on content and format of requests.
FDA final rule effective date is March 7, 2033, when it will assign new 12-digit NDCs and convert all previously assigned 10-digit NDCs for OTC and Rx drugs to the 12-digit NDC format and start a three-year clock for compliance through March 6, 2036.