Politics
After an eventful 2025 that saw the US off-patent industry chalk up wins on tariffs and biosimilar streamlining, AAM president and CEO John Murphy III talks to Generics Bulletin about how the association will be stepping up its lobbying efforts in a US election year.
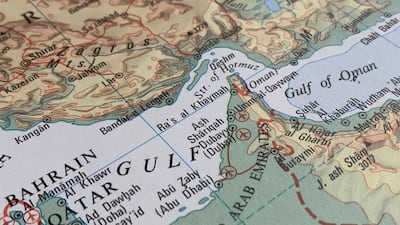
Amid “significant uncertainty” for the off-patent industry caused by the war in Iran, the IGBA has warned that a prolonged conflict “could make solvent supply a critical concern” while escalating costs and squeezed margins could affect the long-term sustainability of affordable medicines.
With concerns building over the potential impact on medicines supply of the war in Iran, Medicines for Europe and Medicines UK have offered a measured assessment of current supply-chain pressures and the potential for future disruption.
As India implements minimum import prices for certain antibiotics and key starting materials, Sandoz's Simon Goeller speaks to Generics Bulletin about the potential for Chinese suppliers to dominate the world market and weaponize supply.
During a candid and revealing discussion, lobbying experts shared trade secrets with the AAM’s Access! 2026 conference on how to get the generics and biosimilars industry’s message heard by lawmakers, as well as looking ahead to the importance of upcoming US midterms.
“The president is certainly hamstrung in many ways by this ruling. For businesses, there's still just sort of a lot of general uncertainty in regard to what else is the president going to try to do, what other authorities might he try to pull,” says Tax Foundation analyst Alex Durante.
Posting action packages for rejected products, not just the complete response letters, is a provocative idea, but also a reminder of what the FDA has lost by not holding many advisory committees during the Trump Administration.
‘This Requires More Than Isolated Policy Interventions’ – Centrient Urges EU To Bolster Supply Chain
Centrient has urged the EU to strengthen its supply chain resilience, including incentivizing local manufacturing through revised procurement practices, after India took action to protect its antibiotics market from an increasing Chinese monopoly.
User fee program changes traditionally are technical and FDA-specific, but the Trump Administration appears to also want policies advocating its “America First” agenda included.
GRAS observations included a report on modernizing FDA published as consumer health industry and food safety advocacy and research groups meet with FDA and OMB officials to offer recommendations for and against a proposed rule agency intends to publish on eliminating self-affirmed GRAS option.
As India implements minimum import prices for certain antibiotics and key starting materials, Sandoz's Simon Goeller speaks to Generics Bulletin about the potential for Chinese suppliers to dominate the world market and weaponize supply.
The clinical and statistical review teams favored a complete response letter for the vasculitis treatment, while the review division and office directors supported approval.