Prescription To OTC Switch
The agency offered to commission a third-party study of efficiency, workload and other factors to determine whether the base PDUFA revenue should be lowered mid-program cycle.
“New Clinical Investigation Exclusivity (3-Year Exclusivity) for Drug Products: Questions and Answers” draft guidance discusses statutory and regulatory criteria for three-year exclusivity eligibility and provides recommendations on content and format of requests.
FDA final rule effective date is March 7, 2033, when it will assign new 12-digit NDCs and convert all previously assigned 10-digit NDCs for OTC and Rx drugs to the 12-digit NDC format and start a three-year clock for compliance through March 6, 2036.
Industry continues to oppose domestic manufacturing incentives that the FDA wants to include in the PDUFA reauthorization.
Including OTC studies in special protocol assessments will be addressed in FDA guidance required in 15 months on improving predictability of process and standards for approval of OTC drug applications, while stepwise labeling information will be post online to improve visibility and searchability fo
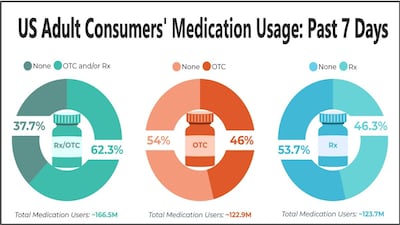
“Observed changes in use prevalence align with changes in regulatory status and availability, including medications” available Rx-only during a 2002 study, “available OTC during the current study period.”
In latest meeting of PDUFA reauthorization nonprescription subgroup, FDAS officials acknowledged stepwise labeling submissions “already available under current regulations but may not be widely known or consistently applied.”
In contributed commentary, Susan Levy, SBL Consulting Group founder and principal, discusses likely health and wellness market developments which would reflect a steady shift toward more accessible, consumer-centric models of care complementing traditional healthcare.
In PDUFA reauthorization subgroup meetings, CHPA suggests expanding FDA’s special protocol assessment program to include OTC drug studies before FDA notes Congress has required it to provide switch-specific guidance before current PDUFA expires.
CDER Office of Generic Drugs publishes MaPP for prescription-to-nonprescription switches and ANDAs to explain regulatory responsibilities for makers of generic copies of reference listed drugs approved for OTC switch.
CDER Office of Generic Drugs publishes MaPP for prescription-to-nonprescription switches and ANDAs to explain regulatory responsibilities for makers of generic copies of reference listed drugs approved for OTC switch.
The tilt toward approval extended beyond patients and advocates to clinicians, professional societies and industry.