Regulatory bodies
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
In an effort to streamline AI rules for high-risk systems and improve competitiveness, two European Parliament committees agreed amendments to the EU AI Act that will allow medtech companies to avoid double certification with notified bodies. A plenary vote takes place in late March.
Smaller biotech companies without the regulatory resources of big pharma should approach the UK medicines regulator and health technology appraisal body for early, informal discussions on how to generate the right evidence.
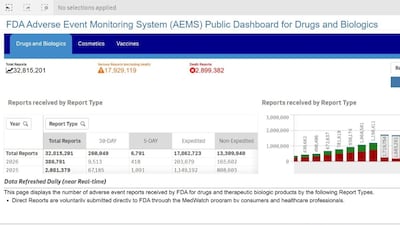
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
The public consultation on the European Commission’s proposed revision of the MDR and IVDR has been extended to accommodate late delivery of translated texts. EU notified bodies’ group TEAM-NB says the proposals need more balance to ensure the resulting regulations do not do more harm than good.
“New Clinical Investigation Exclusivity (3-Year Exclusivity) for Drug Products: Questions and Answers” draft guidance discusses statutory and regulatory criteria for three-year exclusivity eligibility and provides recommendations on content and format of requests.
Regulatory uncertainty has affected business planning and funding opportunities for UK medtech companies, says Gerard Hanratty, head of health and life sciences at UK and Ireland law firm Browne Jacobson. On the other hand, the UK industry should not squander the opportunities it uniquely faces.
Industry groups acknowledge a March 27 public meeting has potential to influence how firms can expand product lines with additional ingredients and formulations allowed under DSHEA. FDA acknowledges industry’s interest in introducing additional ingredients.
Including OTC studies in special protocol assessments will be addressed in FDA guidance required in 15 months on improving predictability of process and standards for approval of OTC drug applications, while stepwise labeling information will be post online to improve visibility and searchability fo
A decade ago, China was buying Western drugs. Now it is selling them. Executives who ignore the shift do so at their peril.
Posting action packages for rejected products, not just the complete response letters, is a provocative idea, but also a reminder of what the FDA has lost by not holding many advisory committees during the Trump Administration.
GRAS observations included a report on modernizing FDA published as consumer health industry and food safety advocacy and research groups meet with FDA and OMB officials to offer recommendations for and against a proposed rule agency intends to publish on eliminating self-affirmed GRAS option.