Retail
“What they want when they think about personalization, is that you meet them on their journey, where they are on their terms. That's what personalization is. How do brands do that? You can't do that by just broadcasting an ad,” CEO Colin Watts says.
Bart Heldreth, Cosmetic Ingredient Review executive director, reflects on the accomplishments of its expert safety panel, which has published safety assessments for more than 7,000 ingredients since CIR’s establishment in 1976.
When House Agriculture Committee on March 3 considers five-farm bill year reauthorization, it will vote whether to add an amendment to extend by a year the Nov. 12 deadline for compliance with lower limits on all THC levels allowed in lawful hemp.
Bills from lawmakers in California (AB 2030) and Connecticut (SB 227) follow similar legislation being considered during current legislative sessions in Alaska, Hawaii, Massachusetts, Michigan and Washington.
“We believe, ultimately, with a personally formulated product, which actually there is, I don't want to say zero competition, but very small competition for, we want to own that category,” says CEO Stephan Gratziani.
Kenvue’s full-year net sales 2.1% dip to $6.38bn came largely on a 2.2% organic sales decline driven by a 2.3% volume decrease but slightly offset by 0.2% foreign currency benefit.
NPA appoints SVP membership; Edible Brands promotes Angela Johnson to CMO; happyactually has medical chief; Nowadays adds reg affairs director; doTerra appoints EVP, innovation; RäFoods names president/CEO; Genesis Lifestyle Medicine adds VP operations; and Hany Salama leads Nutra-Med Packaging.
Returning to full Clear Eyes production and distribution and brand’s previous revenue levels aren’t expected within PCH’s current fiscal year, which ends in March; additional marketing spend expected.
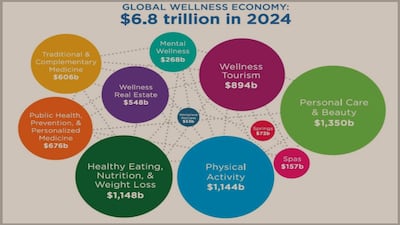
“Wellness real estate has been the leading contributor to wellness economy growth globally, as well as among many high-growth countries that also fall into the top 25 largest markets,” GWI reports.
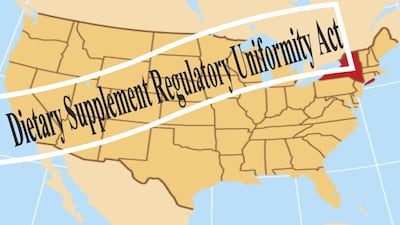
“Dietary Supplement Regulatory Uniformity Act” would prohibit states from adding requirements and rules for supplement manufacturing and sales on top of FDA regulations.
Hawaii proposes age-restricted sales but, unlike similar bills also filed in Alaska, Massachusetts, Michigan and Washington legislatures, would require behind-the-counter storage to limit consumer access in stores.
When FDA warned Agebox about selling iKids-Growth IGF-1 Support supplements as unapproved drugs, agency along with CDC had for around a month been investigating outbreak of infant botulism linked to ByHeart formula.