South America
Moves to issue a compulsory license for Lilly’s GLP-1tirzepatide products Mounjaro and Zepbound come as Brazil prepares to make its own versions of Novo Nordisk’s semaglutide and liraglutide.
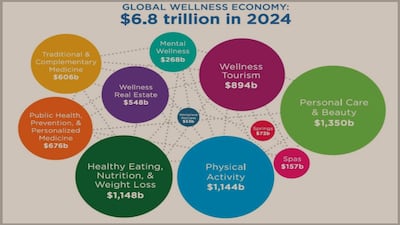
“Wellness real estate has been the leading contributor to wellness economy growth globally, as well as among many high-growth countries that also fall into the top 25 largest markets,” GWI reports.
A biosimilar collaboration between Kashiv and Saya aims to expand access to affordable supportive oncology therapies in Mexico and 10 additional Central America and Caribbean markets.
Two major EU trade agreements are on hold, with ramifications for the pharmaceutical market.
Brazilian authorities say that some pharmaceutical companies exploit court cases brought by patients to secure drug access to achieve higher prices.
Prestige Biopharma has picked Biosidus to manufacture and export its Tuznue biosimilar rival to Herceptin across four Latin American countries.
Argentina’s medicines regulator, ANMAT, is updating the national pharmacopeia with new guidelines on advanced therapies.
Regulatory reliance processes for drug registration and clinical trial application approvals in Brazil have not met their “full potential,” says Brazil’s medicines regulator.
New rules in Argentina mean that good manufacturing practices certificates for foreign drug manufacturing plants issued by certain regulatory authorities could lead to quicker GMP certification evaluations.
A new decree in Brazil establishes a new ethics governance and operational structure in clinical research and seeks to better protect clinical trial participants.
Argentina has implemented a nation-wide traceability system for medicines for the first time.
Brazil is planning to update its National Clinical Research Action Plan to make the country a more competitive location to conduct R&D.