Supply Chain
Bart Heldreth, Cosmetic Ingredient Review executive director, reflects on the accomplishments of its expert safety panel, which has published safety assessments for more than 7,000 ingredients since CIR’s establishment in 1976.
“In a very siloed organization made up of a jigsaw of different companies, no one was thinking enterprise first,” says Perrigo CEO Patrick Lockwood-Taylor. “The fundamentals of running operations, brand-building, innovation, they are now as they were.”
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
At opening session of CHPA’s annual Self-Care Leadership Summit on March 17 in Orlando, it presented Fuad Sawaya its highest honor named for Ivan Combe, an industry pioneer who served on its board from 1958 until 2000.
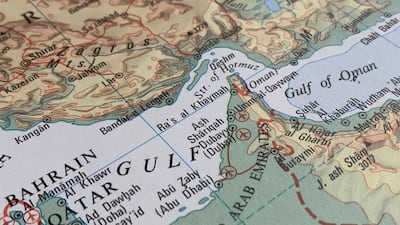
Amid “significant uncertainty” for the off-patent industry caused by the war in Iran, the IGBA has warned that a prolonged conflict “could make solvent supply a critical concern” while escalating costs and squeezed margins could affect the long-term sustainability of affordable medicines.
Speaking to Generics Bulletin on the sidelines of the AAM’s Access! 2026 conference, IGBA chair for 2026 Jim Keon – also president of the CGPA and Biosimilars Canada – discussed IP abuses, supply challenges, biosimilar streamlining and the importance of giving the off-patent industry a global voice.
Antonio Martinez Descalzo has more than 20 years' experience in life sciences, health and nutraceutical industries, including with food and beverage giant ADM. Israeli firm has completed pilot production and is moving into full manufacturing.
With concerns building over the potential impact on medicines supply of the war in Iran, Medicines for Europe and Medicines UK have offered a measured assessment of current supply-chain pressures and the potential for future disruption.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
Campaign launched with video featuring Jang Won-Young away from her glamorous stage image in quiet, everyday moments to reflect the brand's philosophy, "When Skin Heals, You Bloom."
Polpharma has opened an India office to work more closely with API suppliers, improve oversight and strengthen supply chain resilience, marking an early strategic move under new CEO Sebastian Szymanek.
The final week of February saw the AAM hold its Access! 2026 annual conference in Miami and Medicines for Europe hold its annual regulatory and scientific affairs conference in Amsterdam. Generics Bulletin was there at both events and our reporters discuss the key takeaways in our latest podcast.