Surgery
Payers' and patients' growing preference for outpatient care is increasing pressure on private providers to continually reassess how they deliver value. Jean-Philippe Grosmaitre and Guillaume Duparc, partners at L.E.K. Consulting, outlined growth opportunities available to private hospitals.
Surgeon burnout is not a new phenomenon, but where it once was episodic, surgeons now report that it feels more sustained. Support structures have not kept pace, said Johnson & Johnson MedTech VP Africa Wallace on the release of a new study of causes, trends and issues arising.
Surgeon burnout is not a new phenomenon, but where it once was episodic, surgeons now report that it feels more sustained. Support structures have not kept pace, said Johnson & Johnson MedTech VP Africa Wallace on the release of a new study of causes, trends and issues arising.
The EU’s extra layer of checks on clinical evidence is proving a difficult, and sometimes humiliating process, for many device companies whose products have come under the spotlight.
J&J's EMEA head of digital solutions, Julia Fishman, talks about the hurdles in scaling digital innovation and what’s up next on J&J’s innovation road map. Robot-assisted surgery pioneer Ivo Broeders gives his perspective on the difficulties in clinical adoption.
Manufacturing shifts, financial planning, and supply chain changes are among the adjustments that medtech leaders are making to Trump-era tariffs. Medtech Insight’s review of first-quarter earnings calls reveals how firms are navigating the impact and uncertainty of evolving trade policies.
Egg Medical is ramping up efforts to expand international sales of its EggNest scatter radiation protection system to shield X-ray procedure room staff without disrupting workflow. Medtech Insight sat down with Bob Wilson, Egg Medical’s CEO, to discuss the various EggNest systems, marketing opportunities and the competitive landscape.
Tricuspid valve innovation has taken off since the US FDA’s 2023 authorizations of Edwards' Evoque and Abbott's TriClip systems. Whether to repair or replace tricuspid valves remains an open, nuanced question among cardiologists. Dr. Henrik Treede of University Hospital Mainz and TriCares CEO Ahmed Elmouelhi offer views on the evolving space.
This week, Establishment Labs Holdings announced the FDA gave it premarket approval for Motiva breast implant, Cologuard lands FDA approval for Cologuard Plus and GE HealthCare gets FDA nod for a new imaging agent. The FDA announces another expansion for TAP into ophthalmology and radiology. The AAMI and CTA will join forces to develop standards for AI and ML-enabled health care products.
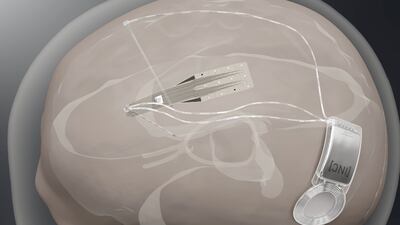
Spain-based Inbrain Neuroelectronics plans first-in-human study to show safety of its graphene-based technology in direct contact with human brain while also developing a second interface for treating Parkinson’s disease.
After announcing positive results showing that its Stentrode BCI is safe in six patients, brain-computer interface company Synchron is planning a pivotal trial to eventually file for FDA approval.
According to Zacks Equity Research, the new trial seeks to support AlphaVac’s adoption in the European market, where pulmonary embolism prevalence and severity is an estimated 435,000 events annually.