Rx-to-OTC Switch
In a foreword to a blog post by PAGB CEO Michelle Riddalls, the UK medicines regulator MHRA has signalled strong support for expanding Rx‑to‑OTC reclassification as a safe way to ease pressure on the NHS, strengthen the role of pharmacists and improve access to medicines.
The agency offered to commission a third-party study of efficiency, workload and other factors to determine whether the base PDUFA revenue should be lowered mid-program cycle.
“New Clinical Investigation Exclusivity (3-Year Exclusivity) for Drug Products: Questions and Answers” draft guidance discusses statutory and regulatory criteria for three-year exclusivity eligibility and provides recommendations on content and format of requests.
Despite 2025 volume stagnation in France and Italy, structural opportunities in category penetration, wellness premiumization, pharmacy expansion, and Rx-to-OTC switch suggest the self-care market still has clear levers for growth.
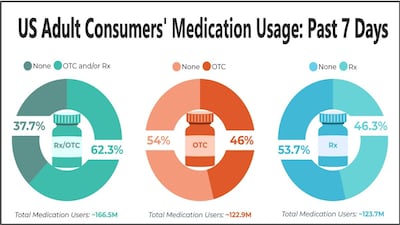
“Observed changes in use prevalence align with changes in regulatory status and availability, including medications” available Rx-only during a 2002 study, “available OTC during the current study period.”
Increased sales of dietary supplements softened France’s 2025 volume decline, which was driven by a mild respiratory season. French self-care industry association NèreS calls for expanded pharmacist authority and broader non‑prescription access to strengthen the country’s first line of care.
Japan is accelerating its tightly managed switch strategy, adding a second emergency contraceptive, expanding EC access to over 7,000 pharmacies, and approving meloxicam as a new pharmacist‑dispensed analgesic.
Germany’s Expert Committee for Prescription (SVA) has unanimously rejected the proposed switch of the controversial painkiller metamizole from prescription to OTC status.
Huge opportunities for OTC therapies loom in India, as consumerization brings structural reset in healthcare. Success stories on Rx-to-OTC transition augur well for pharma, but a predictable regulatory roadmap will be pivotal for sustainable growth.
In PDUFA reauthorization subgroup meetings, CHPA suggests expanding FDA’s special protocol assessment program to include OTC drug studies before FDA notes Congress has required it to provide switch-specific guidance before current PDUFA expires.
Germany’s Expert Committee for Prescription meets on 20 January to discuss the Rx-to-OTC switch of controversial painkiller metamizole, which has been banned in a number of countries around the world because of the risk of agranulocytosis.
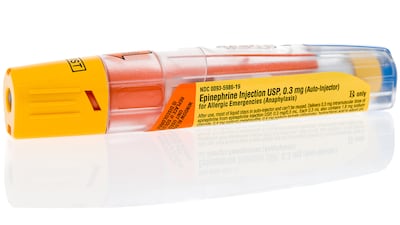
The US FDA is signaling its willingness to consider ‘creative’ proposals to make epinephrine available over-the-counter to treat anaphylaxis. The naloxone OTC switch may be a model, and the new ‘ACNU’ pathway could help.
Agency intends to use information from comments to guide its planning for a public meeting during 2026, according to a Federal Register notice. A GAO report is due in a year to Congress profiling FDA’s progress on making more drugs available nonprescription through OTC switch applications.
UK consumer healthcare industry association, PAGB, suggests improvements that could help accelerate Rx-to-OTC switch in the country.
A new cohort of “techno-enthusiasts” who are highly susceptible to misinformation and inappropriate self-care decisions are undermining Italy's responsible self-medication model, finds a report by Italian self-care industry association, Assosalute, and independent researcher Censis.
Negative growth in global OTC market sales volume means that “as an industry, we're not doing good enough,” argues Opella’s Asia Pacific, Middle East and Africa (AMEA) head, Carol-Ann Stewart, in this exclusive feature interview.
Approval of numerous Rx formulations hasn’t translated to wider availability when a remedy is needed for anaphylaxis emergency, say FDA and the Margolis Institute in announcing workshop on reducing anaphylaxis-related morbidity and mortality.
Self-Care Week Special: HBW Insight catches up with PAGB CEO Michelle Riddalls OBE to talk about what the association is doing to promote Rx-to-OTC switch in the UK.
The global allergy market is “expanding again but on new terms, driven by a mix of regional strength, evolving consumer behaviors, and an increasingly diverse competitive field,” according to the latest IQVIA Consumer Health analysis.
Japan’s Ministry of Health, Labor and Welfare continues to widen access to non-prescription medicines with another Rx-to-OTC switch – Rohto Algard Epinastine Eye Drops – and a direct OTC approval of Sato Pharmaceutical's Menofeminin (black cohosh) menopause treatment.