Diabetic Care
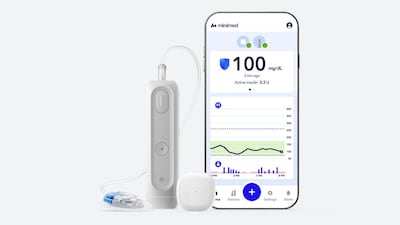
After its IPO, MiniMed has its first FDA clearance as a public company. The MiniMed Flex – smaller, screenless, and smartphone-controlled – is the product the newly independent diabetes firm needed to shift the conversation from market debut to market execution.
A turbulent market backdrop, geopolitical shock following the US and Israel striking Iran days before pricing, and a limited public float is likely to have weighed on investor appetite.
Medtronic plans to retain about 90% of the newly standalone MiniMed. While MiniMed’s 2025 revenues totaled $2.7bn, the company still posted a net loss amid strong competition from Abbott and Dexcom and recently announced more than six dozen layoffs at its California plant.
The US FDA says incorrect code messages from some Trividia glucose measurement devices could result in delayed treatment and serious patient harm.
The US FDA hosted a virtual town hall as a follow-up to final guidance the agency issued last month, which outlines its policy on general wellness medical devices that the agency considers low risk.
A warning letter from the US FDA citing concerns of some Abbott continuous glucose monitors will not stop the company from launching a novel diabetes sensor later this year as planned.
BioStem Technologies’ buyout of BioTissue Holding’s surgical and wound-care business adds cryopreserved and sterile technologies, Cryotek and SteriTek, and a direct sales force focused on acute care settings.
Withings announced at CES that its health monitoring app will integrate data from Abbott’s OTC CGM Lingo starting in Q1.
Timed to debut during this week’s CES 2026 technology conference in Las Vegas, the new feature within Abbott’s Libre glucose monitoring software uses generative artificial intelligence to predict how food choices may affect glucose levels.
Last year was a busy one for the medtech industry, with major advances in technology as well as big changes at regulatory agencies. Get a peek at the stories our readers couldn't miss.
MiniMed flagged launch-related challenges in its IPO filing, saying production of its Simplera CGM is scaling more slowly than expected due to underperformance in initial high-volume automated manufacturing.
The system combines a durable rechargeable transmitter with a two-year life span and a replaceable single-day sensor. The product is intended to address pain, inconvenience and adherence challenges associated with multiweek CGM use.
The US FDA is launching a pilot to promote access and safety to digital health devices. Developed by the agency’s device center, the pilot will evaluate a new, risk-based enforcement approach for certain types of digital devices to treat several conditions.
New rule in effect Jan. 1 follows OIG call for CMS to use competitive bidding to adjust payments for continuous glucose monitors after finding that Medicare was paying above supplier costs and retail prices.
US Medicare payments for CGMs and supplies in the year to June 2023 exceeded suppliers’ acquisition costs by $377m, says new OIG report
Abbott has initiated a medical device correction for about 3 million FreeStyle Libre 3 and FreeStyle Libre 3 Plus sensors in the US after internal testing detected a risk the device may produce inaccurate glucose readings.
Brain-computer interfaces advance toward trials and commercialization, Oura pushes for FDA-cleared blood pressure monitoring, and regulators weigh AI’s expanding role in mental health and diagnostics amid rising safety concerns.
Tandem Diabetes Care is positioning for growth in 2026 with the launch of its Mobi Tubeless pump following FDA clearance of its Android-compatible Mobi app while navigating multiple recalls during what CEO John Sheridan called a transformative year.
Viome’s CEO To Expand Into Clinical Diagnostics ‘For Which There Are No Solutions’ With AI, RNA Test
Viome Life Sciences accelerates its push into clinical diagnostics with studies in colorectal cancer and a major partnership with Microsoft to scale its molecular data analysis platform. Viome leverages RNA analysis and AI to detect disease at the molecular level and personalize preventive health.
Wearables innovator Oura has launched a profile study aimed at helping the company secure FDA clearance for a blood pressure feature for its smart ring. Study participants will answer questions while wearing the ring, which the company plans to use to support the feature’s efficacy.