Trump 2.0
The threat of Chinese dominance in the prescription drug sector appears to be a strong, unifying theme for legislators heading into the US Food and Drug Administration user fee reauthorization cycle, but two very different sets of policy issues must be addressed.
Payroll costs in the prescription drug user fee program increased in fiscal year 2025 despite the reduction-in-force and voluntary departures, which could impact industry efforts to reduce the annual charges.
After an eventful 2025 that saw the US off-patent industry chalk up wins on tariffs and biosimilar streamlining, AAM president and CEO John Murphy III talks to Generics Bulletin about how the association will be stepping up its lobbying efforts in a US election year.
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
The US FDA will seek industry feedback on the Commissioner’s National Priority Voucher pilot during a June public meeting as the ultra-fast drug review program marks its first year amid growing scrutiny of its scope, timelines, and sponsor obligations.
In a signal that prescription drug and generic drug user fee talks are nearing completion, the FDA and industry are preparing to start biosimilar user fee reauthorization talks with America First and other issues potentially looming.
Pink Sheet editors discuss the impact of a federal judge’s decision that the recent ACIP membership turnover and vaccine schedule changes likely violated statutes, as well as the FDA’s look at new opioid disposal requirements.
With the US CDC Advisory Committee on Immunization Practices sidelined by the courts, the Pink Sheet imagined how a past version of the panel may have spent the spring, including tracking a fast‑moving pipeline of flu, RSV, Lyme, dengue, and next‑generation combination vaccines.
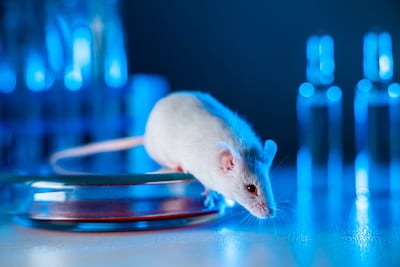
Context of use, human biological relevance, technical characterization and fitness for purpose are the key elements in validating novel methods to replace animal testing, the FDA said in a new draft guidance.
The agency is conducting further investigation of reports of altered skin sensation that occurred more frequently with the 7.2 mg semaglutide dose than with lower doses of the GLP-1 inhibitor.
The agency offered to drop annual IND fees for sponsors not conducting Phase I trials in the US, but industry still raised several concerns about fee incentives for onshoring clinical development.
A US lawmaker described program reform goals as returning the program to its original focus on supporting federally qualified health centers and rural hospitals.