Surgery
Neurent’s Neuromark device, which relieves symptoms of chronic rhinitis via ablation of the vidian nerve, could be well-poised to capture an ever-growing and highly valuable market.
Numares Health bets on a metabolomics-based urine test to help doctors detect kidney transplantation rejection earlier than current biomarkers, which would allow doctors to intervene earlier in trying to save the transplanted organ.
The US FDA has issued an early alert concerning Intuitive Surgical staplers used during various procedures with the da Vinci Surgical System. The company is instructing customers to stop using the devices.
Paradromics has launched a new academic-industry collaboration with leading brain-computer interface researchers at Stanford, Mass General, Pitt, UC Davis and Michigan aiming to translate BCI research into devices to help people with speech impairment, stroke and Parkinson’s.
SS Innovations said the net proceeds from the financing will be directed toward working capital and general corporate purposes, with a focus on advancing global expansion efforts across existing and target markets.
Surgeon burnout is not a new phenomenon, but where it once was episodic, surgeons now report that it feels more sustained. Support structures have not kept pace, said Johnson & Johnson MedTech VP Africa Wallace on the release of a new study of causes, trends and issues arising.
The US FDA has issued an early alert concerning Abiomed’s Impella purge cassettes, a critical heart pump component that delivers rinsing fluid to the Impella catheter.
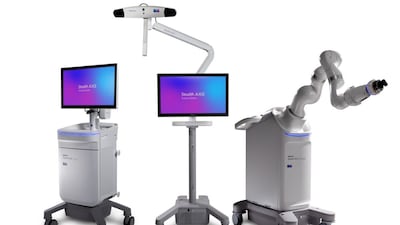
The Stealth AXiS system is designed as an integrated platform that combines robotics, surgical planning and navigation into a single solution for spine procedures.
While surgical technologies is a roughly $6bn Medtronic business with “various moving parts,” Hugo is not yet large enough to materially shift surgical performance, it is growing at a strong pace, CEO Geoff Martha said.
The medical device industry continues to face a paradox: Despite substantial investments in research and development, many innovative technologies fail to gain traction with surgeons. This article explores the multifaceted drivers of surgeon adoption behavior and provides actionable insights.
The medtech giant violated USA and California antitrust law in its sales practices for devices used to cut and seal blood vessels during surgery, a jury found. Rival firm Applied Medical brought the case.
Stryker’s broader orthopedics portfolio continued to benefit from strong robotic demand during the quarter, with the company reporting 28.7% organic growth in its US Orthopedics other than hips and knees business.
Medtronic has agreed to acquire Cathworks for up to $585m, converting a long-standing partnership into full ownership as it expands its footprint in coronary physiology and AI-enabled cardiovascular tools.
The US FDA says Abiomed has reported 22 serious injuries linked to an issue with some of its temporary heart pumps that provide support to patients with acute right heart failure.
INBRAIN unveiled a bidirectional "rice-sized" BCI chip partnership, Merck commercialization progress and new speech-decoding trial in France as it advances its graphene-based cortical interface toward commercialization, pending regulatory clearance.
In this final part of a three-part series, Medtech Insight spoke with a neuroethicist and the first person in a trial using a BCI implant for stimulating hand movement. This story explores ethical considerations that arise when projects can no longer support patients with implanted devices.
The FDA has cleared Intuitive Surgical’s da Vinci 5 for some cardiac procedures, opening new opportunities for the firm. The company is also looking to expand its presence in ambulatory surgical centers, execs said during a Jan. 22 earnings call.
BioStem Technologies’ buyout of BioTissue Holding’s surgical and wound-care business adds cryopreserved and sterile technologies, Cryotek and SteriTek, and a direct sales force focused on acute care settings.
Abbott’s newly CE-marked TactiFlex Duo dual-ablation catheter used for treating AFib is competing against products already introduced by Boston Scientific, J&J and Medtronic.
The US FDA has issued an early alert concerning some Axios stent and delivery systems used in endoscopic drainage procedures after receiving reports of serious injuries and deaths linked to the device.