Neurological
Deal snapshot: Preparing for submission and potential approval and launch of its Phase III sleep apnea drug, Apnimed is divesting its half of Shionogi-Apnimed Sleep Science for $100m.
For $650m up front, Collegium will pair Azstarys, which provides both immediate and extended onset of action, with Jornay PM, its ADHD drug indicated for bedtime dosing.
As Spinraza’s sales have slumped, Biogen is developing the once-yearly salanersen, showing efficacy in patients with suboptimal responses to Novartis’s Zolgensma.
Phase III results for azetukalner in focal onset seizures bested the impressive placebo-adjusted Phase IIb data for the first-in-class drug, which Xenon designed to overcome challenges of other anti-seizure medications.
The Phase III anti-alpha-synuclein antibody has the potential to become a first-in-class therapy for multiple system atrophy.
At BIO’s Investor and Growth Summit, investors acknowledged the tough funding environment for early stage neuroscience, but said there are signs of increasing interest in the space.
Roche believes its drug has a strong benefit-risk profile but analysts think the eight trial deaths could see it rejected, just like Sanofi’s rival BTK inhibitor.
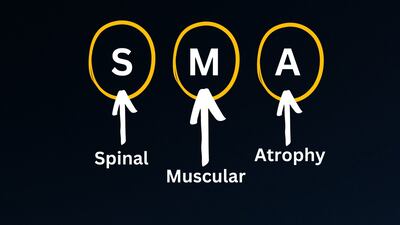
The controversial Doug Ingram will depart for personal reasons, following the diagnosis of two family members with myotonic dystrophy.
The company’s next VC round hinged on Phase I results for QRL-201, an antisense oligonucleotide designed to restore STMN2 expression in ALS patients, and topline data are promising.
Emerging from stealth mode, Slate has revealed an in licensed anti PACAP migraine program, entering a field that includes efforts from Lundbeck.
AC Immune said J&J paused enrollment in the Phase IIb RETAIN study of ACI-35.030/JNJ-2056 in preclinical Alzheimer’s disease to evaluate aspects of the trial, including recruitment.
2026 has already seen some interesting late-stage trial readouts, from Sanofi’s anti-OX40L antibody, amlitelimab, in atopic dermatitis to Vertex’s APRIL/BAFF inhibitor, povetacicept, in IgA nephropathy. Here Scrip surveys of some of the more interesting Phase III clinical trials still to come.
2026 has already seen several major new funding rounds for Chinese biopharma ventures, including a $70m financing for CAR-T specialist Oricell and a $40m Angel round for ADC specialist Tosun.
Positive Phase IIb data positions Lundbeck’s anti-PACAP antibody bocunebart for Phase III discussions, potentially expanding the company’s migraine franchise beyond its CGRP therapy Vyepti.
The Irish firm has raised a total of $60m to develop its antibody-oligonucleotide conjugate platform.
After more than a decade developing its nanoparticle radioenhancer technology, Nanobiotix and partner Johnson & Johnson believe it could soon begin to revolutionize frontline oncology treatment.
The company hopes to prove its RNAi platform is the most versatile and potent in cardiometabolic therapies in 2026 – and win one over on its long-standing rival, Ionis.
Enthusiastic predictions from industry leaders for ongoing progress in obesity, oncology, immunology, neuroscience and more were supported by optimism for the potential of AI and other technological advances to enable breakthroughs in processes as well as pipelines.
The drugmaker highlighted a wide array of development programs in its fourth quarter earnings, including moving tirzepatide into multiple inflammatory conditions.