United States
During an ECRI-hosted webinar on AI-enabled diagnostics, a panel of experts discussed the promise and risks that come with the technology for both physicians and patients alike.
Small Biz Bill A Big Win For Medtech Innovation: Bipartisan Legislation Awaits President’s Signature
The US Congress overwhelmingly passed legislation reauthorizing federal programs essential to smaller medtech firms in need of funding. AdvaMed says the bill is critical for medtech innovation.
The FDA approved JenaValve’s Trilogy HeartValve for treating severe aortic regurgitation, marking it as the first TAVR device for this condition in the US. The approval, which followed promising clinical results, is significant for patients with limited options due to high surgical risks.
The US FDA has issued an early alert concerning Intuitive Surgical staplers used during various procedures with the da Vinci Surgical System. The company is instructing customers to stop using the devices.
A recent FDA report to Congress reflects ongoing safety issues with orthopedic and cardiac implants, as well as commodities such as syringes and PPE.
The Trump EPA wants to loosen restrictions the Biden administration placed on facilities that sterilize medical devices with ethylene oxide, arguing the rollbacks are necessary to ensure device accessibility and supply chain security. Opponents to the rollbacks say EtO is a danger to public health.
A new final guidance document from the US FDA lays out premarket considerations for devices intended to help treat obesity, including clinical trial design, labeling, and safety concerns. The guidance also calls for sponsors to consider patient perceptions in evaluating device effectiveness.
During the latest round of MDUFA negotiations, the FDA and medtech industry agreed to include language in the next commitment letter that would provide direction for the use of carryover funds from user fees. The parties also found agreement on several other topics.
As AI becomes ubiquitous in diagnosing diseases, clinicians must balance the technology’s enormous upside, such as improving the speed and accuracy of diagnoses, with the potential for errors that can lead to delayed or missed treatment.
The FDA’s device center has received a second citizen petition relating to the NarxCare algorithm by Bamboo Health. Both petitions argue that NarxCare qualifies as a regulated clinical decision support product, but it is not clear whether the second petition will succeed where the first failed.
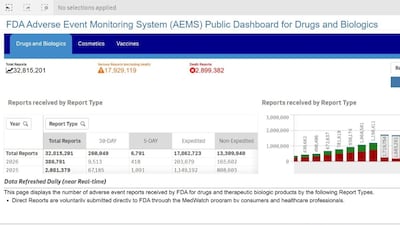
The US FDA is replacing its individual product type adverse event reporting portals with a unified database, known as AEMS. While adverse event reports for some sectors such as drugs and vaccines joined the new portal on March 11, devices, human foods and tobacco products will be added in May.
ExThera Medical is facing legal trouble after two patients died following treatment with its blood filtration device. The company's former chief regulatory officer has pled guilty to failing to report adverse events, while ExThera signed a deferred prosecution alert and agreed to pay $6.45m.
HeartBeam’s CEO says the strength of clinical data convinced the US FDA to reverse course and grant clearance to his company’s innovative ECG device, which he says can assess arrhythmias at home with hospital accuracy.
Recently published lists show that the US Food and Drug Administration had cleared almost 2,000 digital health tools by the end of 2025. Of these, well over half – 1,104 – were radiology devices incorporating AI. Find a full breakdown in these tables.
During MDUFA negotiations held Feb. 11, the medtech industry agreed to a proposal from the FDA to base its hiring commitments on MDUFA IV.
Real-world evidence (RWE) is increasingly significant in medical device submissions, with FDA guidance evolving to embrace RWE benchmarks. The National Evaluation System for Health Technology (NEST) partners with firms to streamline RWE use, enhancing regulatory processes.
CMS is asking stakeholders to help the agency craft potential regulatory changes aimed at bolstering its efforts to combat Medicare and Medicaid fraud. The agency has provided several topics to help guide stakeholder engagement.
CMS has reopened the TAVR coverage memo to consider coverage for without symptoms aortic stenosis and pure regurgitation. Stakeholders want to relax CED requirements and update NCD to include newer FDA-approved TAVR devices, improving patient access and outcomes in valvular heart disease treatment.
The Trump administration says a six-month moratorium on enrolling new suppliers of durable medical equipment is part of a wider strategy to combat healthcare fraud and abuse.
The US Medicare agency is now covering superficial radiotherapy (SRT) for the treatment of skin cancer. Joseph Sardano, CEO of SRT firm Sensus Healthcare, spoke to Medtech Insight about the potential of the technology and why it’s still facing adaptation challenges.