Compliance
During an ECRI-hosted webinar on AI-enabled diagnostics, a panel of experts discussed the promise and risks that come with the technology for both physicians and patients alike.
In an effort to streamline AI rules for high-risk systems and improve competitiveness, two European Parliament committees agreed amendments to the EU AI Act that will allow medtech companies to avoid double certification with notified bodies. A plenary vote takes place in late March.
The Trump EPA wants to loosen restrictions the Biden administration placed on facilities that sterilize medical devices with ethylene oxide, arguing the rollbacks are necessary to ensure device accessibility and supply chain security. Opponents to the rollbacks say EtO is a danger to public health.
A new final guidance document from the US FDA lays out premarket considerations for devices intended to help treat obesity, including clinical trial design, labeling, and safety concerns. The guidance also calls for sponsors to consider patient perceptions in evaluating device effectiveness.
The FDA’s device center has received a second citizen petition relating to the NarxCare algorithm by Bamboo Health. Both petitions argue that NarxCare qualifies as a regulated clinical decision support product, but it is not clear whether the second petition will succeed where the first failed.
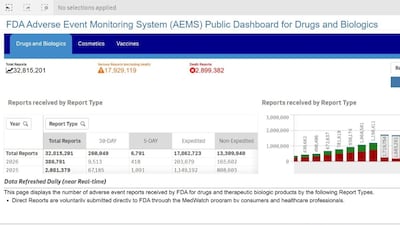
The US FDA is replacing its individual product type adverse event reporting portals with a unified database, known as AEMS. While adverse event reports for some sectors such as drugs and vaccines joined the new portal on March 11, devices, human foods and tobacco products will be added in May.
ExThera Medical is facing legal trouble after two patients died following treatment with its blood filtration device. The company's former chief regulatory officer has pled guilty to failing to report adverse events, while ExThera signed a deferred prosecution alert and agreed to pay $6.45m.
Stay current on regulatory guidelines from around the world with Medtech Insight's Guidance Tracker. Over 30 documents have been posted on the tracker since its last update.
The US FDA is warning that anti-choking devices can prove ineffective in emergencies and that methods established by the American Red Cross and the American Heart Association to remove blockages from choking victims should be tried first.
In two warning letters posted Feb. 24, the US FDA cited a device manufacturer with clinical trial violations and another for failing to meet current good manufacturing practices.
US FDA staff discussed a newly in effect guidance document on the use of real-world evidence in medical device submissions in a recent webinar. The new guidance broadens the set of data sources the agency will accept, while also clarifying the FDA’s expectations for sponsors using RWE.
Class I medical device recalls reached record-breaking levels in 2025, according Sedwick’s annual product safety report, with device failure the leading cause.
The US FDA hosted a virtual town hall as a follow-up to final guidance the agency issued last month, which outlines its policy on general wellness medical devices that the agency considers low risk.
The US FDA has published an updated inspections manual detailing how it will conduct investigations going forward under its new regulatory framework. Medtech Insight talked to a couple of regulatory experts about the key changes to the new protocol.
Recent MDUFA VI negotiations emphasized real-world evidence and potential reforms in de novo and presubmission programs. Both industry representatives and FDA staff discussed improving RWE quality and addressing device submission challenges.
Twenty-one companies received US FDA warning letters in December and January. About half were part of a crackdown on unregistered vendors of breast binders, while other recipients included Abbott and Magellan Diagnostics.
Stay current on regulatory guidelines from around the world with Medtech Insight's Guidance Tracker. Over 40 documents have been posted on the tracker since its last update.
A warning letter from the US FDA citing concerns of some Abbott continuous glucose monitors will not stop the company from launching a novel diabetes sensor later this year as planned.
Recommendations to mitigate attrition among clinical research and drug manufacturing investigators at the FDA have not been implemented, and a Strategic Workforce Plan aimed at addressing recruiting, retention and training challenges has been shelved, the Government Accountability Office said.
The US FDA has warned that Magellan Diagnostics blood lead tests may provide inaccurately high results, particularly with tubes from ASP Global. Magellan’s tests had previously been recalled over inaccurate low results. The FDA also issued warning letters to both firms.