Legislation
The threat of Chinese dominance in the prescription drug sector appears to be a strong, unifying theme for legislators heading into the US Food and Drug Administration user fee reauthorization cycle, but two very different sets of policy issues must be addressed.
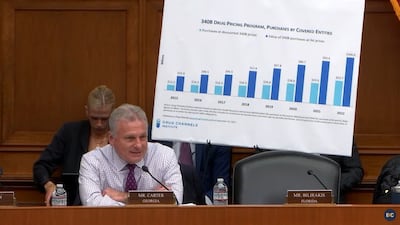
A US lawmaker described program reform goals as returning the program to its original focus on supporting federally qualified health centers and rural hospitals.
The draft report lays out a unified EU framework to reduce disparities and modernize rare disease diagnosis, treatment, care and research.
India issues firm advisory on promos for obesity and metabolic disorders therapies ahead of loss of exclusivity for semaglutide later this week, with 50-plus generic brands seen in the wings for a share of the pie. Will Eli Lilly and Novo Nordisk have to tweak their multimedia campaigns?
Leem, the French industry association, is reaching out to patients, carers and the general public with the aim of building better policies to improve access to medicines and ensure the pharmaceutical industry is competitive.
The pharmaceutical industry now has clarity on exactly how the new framework for governing medicinal products in the EU will work, with changes ahead for regulatory data protection, novel antimicrobials and orphan medicines.
The overhaul of the EU pharmaceutical package is expected to introduce two new regulatory “concepts” relating to platform technologies. A European Commission expert explains how the changes could impact industry and what the expected benefits are.
During a candid and revealing discussion, lobbying experts shared strategies for getting the generics and biosimilars industry’s message heard by lawmakers.
During a Senate hearing, rare disease stakeholders said the FDA is not engaging with patients as much as before and suggested it may be hindering drug development.
Legal procedures brought by Poland and Ireland could still force a rethink of controversial measures in the updated Urban Wastewater Treatment Directive, despite a recent legal setback for the pharmaceutical industry.
Trump touted his "big achievement" on drug pricing during his State of the Union address and did not imply other reforms are coming.
AAM President and CEO John Murphy summarized a year of industry progress in front of generics and biosimilars industry delegates at AAM’s Access! 2026 conference, but also said more efforts were needed to create a sustainable future environment.
Findings from a new report on electronic Product Information pilots projects in Europe reveal a landscape of innovation, collaboration and cautious optimism, according to a group of pharmaceutical industry bodies.
The FDA’s Grace Graham and Kyle Diamantas, and CMS’ Chris Klomp and John Brooks, will retain their respective agency leadership roles as well as manage the agencies’ issues at HHS, with healthcare likely a major issue in the upcoming congressional campaign.
During a House Energy and Commerce Health Subcommittee hearing, chair Morgan Griffith suggested he would support efforts to learn more about the Most Favored Nation deals between the Trump Administration and individual drug companies.
Pharma firms are keen to ensure that artificial intelligence tools used in the earlier stages of drug discovery are excluded from the “onerous scope” of the EU’s AI Act, as this could “defeat” the benefits of using this technology, says Clifford Chance’s Stephen Reese.
The Trump administration has launched TrumpRx, a website the White House says could help save Americans billions in pharmaceutical spending, although it does not itself sell or dispense drugs.
The US Labor Department also added a proposed rule requiring PBMs to disclose additional compensation information to certain employer-plan sponsors.
If the EU’s controversial Artificial Intelligence Act applies restrictive rules to pharmaceutical research and development activities, the UK could benefit from offering a more favorable regulatory environment, top lawyer Stephen Reese explains.
Patients severely burned in a Swiss bar fire have been treated with a novel, life-saving antimicrobial containing sulbactam and durlobactam. But this drug has never been filed for approval in the EU, something that is “disappointing” according to the EMA’s Marco Cavaleri.