Policy & Regulation
FDA aims to prevent poor quality or incomplete responses to Form 483 inspection observations with a new draft guidance describing the structure and content for concise, factual and effective corrective action responses.
Brazil has approved legislation permitting pharmacies to operate within supermarkets, expanding consumer access to OTC medicines while maintaining strict pharmacist oversight and existing safety standards.
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
Context of use, human biological relevance, technical characterization and fitness for purpose are the key elements in validating novel methods to replace animal testing, the FDA said in a new draft guidance.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
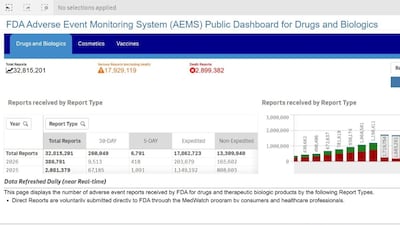
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
The second panel of this week's Geneva Self‑Care Forum explored further the tension between self‑care as a public‑health tool and self‑care as an out‑of‑pocket consumer market, raising sharp questions about where the industry sits.
The global consumer health industry sees emerging markets as an engine for growth in an otherwise stagnating market. But panelists at the Global Self-Care Forum in Geneva warned that regulatory gaps and lack of harmonization may stunt this potential in the short term.
FDA final rule effective date is March 7, 2033, when it will assign new 12-digit NDCs and convert all previously assigned 10-digit NDCs for OTC and Rx drugs to the 12-digit NDC format and start a three-year clock for compliance through March 6, 2036.
Self-care is gaining momentum worldwide, as the Global Self-Care Federation Forum in Geneva made clear. But a central question remains: if self-care is to deliver on its promise of healthier populations and more sustainable healthcare systems, who will finance it?
Industry continues to oppose domestic manufacturing incentives that the FDA wants to include in the PDUFA reauthorization.
In this episode of HBW Insight’s Over the Counter podcast, we speak with probiotic expert David Pineda Ereño about Europe’s fragmented regulatory landscape, the future of the “probiotic” definition, and how companies can navigate diverging EU Member State positions.
Including OTC studies in special protocol assessments will be addressed in FDA guidance required in 15 months on improving predictability of process and standards for approval of OTC drug applications, while stepwise labeling information will be post online to improve visibility and searchability fo
With EFPIA’s challenge struck out, the UWWTD battle now shifts to Poland’s case at the Court of Justice, where AESGP expects the Directive’s core legal arguments to finally be tested.
Japan is tightening access to eight widely used OTC ingredients from May 2026 to curb youth misuse, a move broadly accepted by industry but expected to constrain online availability due to strict face‑to‑face or video‑verified sales requirements.
Findings from a new report on electronic Product Information pilots projects in Europe reveal a landscape of innovation, collaboration and cautious optimism, according to a group of pharmaceutical industry bodies.
Despite 2025 volume stagnation in France and Italy, structural opportunities in category penetration, wellness premiumization, pharmacy expansion, and Rx-to-OTC switch suggest the self-care market still has clear levers for growth.
Increased sales of dietary supplements softened France’s 2025 volume decline, which was driven by a mild respiratory season. French self-care industry association NèreS calls for expanded pharmacist authority and broader non‑prescription access to strengthen the country’s first line of care.
User fee program changes traditionally are technical and FDA-specific, but the Trump Administration appears to also want policies advocating its “America First” agenda included.
“Your firm’s quality systems are inadequate” or “these products are unapproved new drugs” each firm warned with reference to FDA’s regulation for GMPs for pharmaceuticals, supplements or cosmetics. Warnings included 10 to firms about GMP violations topical drugs production.