Business
With its new parntership with Colorado's Verb Biotics, Opella says it wants to create “clinically proven gut health solutions, ready for real life.”
In Part 2 of HBW Insight’s Over the Counter podcast with David Pineda Ereño, we discuss how consumer health companies are navigating fragmented EU rules and leveraging e‑commerce to exploit the commercial potential of postbiotics. We also briefly discuss the exciting area of postbiotics.
AHPA Awards Hero, Champion, Visionary and others at annual meeting; Kimberly-Clark’s information chief has major-merger experience; RäFoods’ marketing chief has history with CEO; and Eshbal adds VP for North America expansion.
VALR Energy names appoints chief revenue officer; CEO at Audien Hearing; Anderson retiring at NACDS; Lindora appoints president and chief medical officer; and USHR expands leadership.
Innovation & IP
Just a 1% improvement thanks to probiotic use in areas like antibiotic‑associated diarrhea, respiratory tract health, oral health, women’s health, lactose digestion, and mental wellbeing could generate significant savings for Europe's strained health systems.
Antonio Martinez Descalzo has more than 20 years' experience in life sciences, health and nutraceutical industries, including with food and beverage giant ADM. Israeli firm has completed pilot production and is moving into full manufacturing.
With its new parntership with Colorado's Verb Biotics, Opella says it wants to create “clinically proven gut health solutions, ready for real life.”
In Part 2 of HBW Insight’s Over the Counter podcast with David Pineda Ereño, we discuss how consumer health companies are navigating fragmented EU rules and leveraging e‑commerce to exploit the commercial potential of postbiotics. We also briefly discuss the exciting area of postbiotics.
Policy & Regulation
In Part 2 of HBW Insight’s Over the Counter podcast with David Pineda Ereño, we discuss how consumer health companies are navigating fragmented EU rules and leveraging e‑commerce to exploit the commercial potential of postbiotics. We also briefly discuss the exciting area of postbiotics.
US Trade Representative is taking two swings under Section 301(b) of the 1974 Trade Act. Customs and Border Protection has March 19 to provide US Court of International Trade with next update on its progress on providing refunds for IEEPA tariffs.
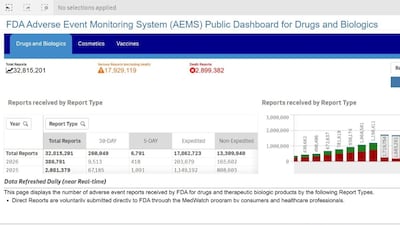
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
FTC’s sending checks totaling more than $40,700 to 578 consumers who purchased deceptively marketed treatment plans from Golden Sunrise between July 2017 and July 2020. Company owner and chief medical officer both pleaded guilty to criminal charges.