Regulation
FDA aims to prevent poor quality or incomplete responses to Form 483 inspection observations with a new draft guidance describing the structure and content for concise, factual and effective corrective action responses.
Agency offered to drop annual IND fees for sponsors not conducting Phase I trials in US, but industry still raised several concerns about fee incentives for onshoring clinical development.
Context of use, human biological relevance, technical characterization and fitness for purpose are the key elements in validating novel methods to replace animal testing, the FDA said in a new draft guidance.
FY 2026 OMUFA facility fees are $19,188 for OTC monograph drug manufacturers and $12,792 for contract manufacturing organizations also making the products. FY 2025 fees were $37,556 and $25,037.
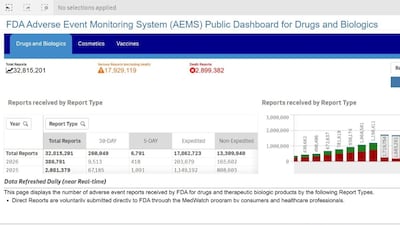
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
FDA final rule effective date is March 7, 2033, when it will assign new 12-digit NDCs and convert all previously assigned 10-digit NDCs for OTC and Rx drugs to the 12-digit NDC format and start a three-year clock for compliance through March 6, 2036.
Findings from a new report on electronic Product Information pilots projects in Europe reveal a landscape of innovation, collaboration and cautious optimism, according to a group of pharmaceutical industry bodies.
In latest meeting of PDUFA reauthorization nonprescription subgroup, FDAS officials acknowledged stepwise labeling submissions “already available under current regulations but may not be widely known or consistently applied.”
FDA updates pair of guidance documents relaxing its posture on how it regulates general wellness devices and clinical support software. Industry mostly welcomes changes, agreeing with agency’s view that lighter regulation will help spur innovation.
“Our sense is that companies are currently focused on timing, scope and execution. For example, knowing when a final order might issue … We also imagine that formulators are scrutinizing the proposed list of permitted combinations, the 6% cap, and the permitted dosage forms,” say ArentFox Schiff att
Agency says FY 2026 Tier 1 OMOR fees are $587,529 and Tier 2 fees are $117,505. The fees, due to FDA when an OMOR is filed, aren’t included in OMUFA target revenue calculation for each fiscal year based entirely on annual facility registration fees.
Former agency officials now representing industry worry that a deregulatory bent could be driving "Simple Reform" plan to merge all medical product and clinical research inspectorates and that specialist expertise gained in 2017 "Program Alignment" initiative will be reversed.
Sunscreen products industry and public health advocacy groups have been critical that FDA has not approved a new filter since 1999 even as countries in Europe and other regions allow using numerous additional ingredients in sunscreens.
Negative growth in global OTC market sales volume means that “as an industry, we're not doing good enough,” argues Opella’s Asia Pacific, Middle East and Africa (AMEA) head, Carol-Ann Stewart, in this exclusive feature interview.
Scientific literature on potential link between prenatal acetaminophen exposure and neurological conditions “now includes FDA’s most recent epidemiological review” completed in May saying research results “insufficient to support a causal association.”
Norway's Medical Products Agency has rejected claims that misuse of OTC ibuprofen is linked to its availability for purchase in outlets other than pharmacies.
BRS Analytical Services in St. Louis recalled more than 75,000 cases of redness and lubricating eye drops and committed “to permanently cease production of all drugs.”
The “Simple Reform” initiative would reverse a 2017 move to ensure FDA investigators were experts in the commodity they inspected or in clinical research regulations.
Informed Consent Action Network requests warning on acetaminophen-containing OTC monograph drugs in petition submitted by Siri & Glimstad LLP, where partner Aaron Siri has provided legal counsel for HHS Secretary Kennedy. Tylenol marketer Kenvue disputes ICAN’s conclusions and its grasp of FDA regul
Panelists at the BioFuture conference discussed the potential opportunities the government shutdown creates for the FDA to improve flexibility and efficiency.