Policy & Regulation
The Expert Panel for Cosmetic Ingredient Safety wants data to better understand airbrush cosmetic products and devices, but does not expect it will receive it.
Circular Action Alliance, the Producer Responsibility Organization in charge of implementing California’s EPR program, urges single-use packaging producers to comply with an upcoming May 31 deadline, which will shape proposed fees.
President Trump issued an Executive Order on ‘Made in America’ and similar US-origin claims that will likely result in increased FTC enforcement on marketing with the claims and the potential for increased pressure on online marketplaces.
Johnson & Johnson’s petition for a writ of certiorari with the US Supreme Court has drawn the attention of the US Chamber of Commerce and other trade associations, which have filed an amicus brief in support of the firm.
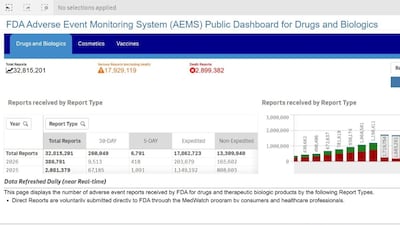
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
Following a final 'unsafe' determination on prostaglandin analogues from the Scientific Committee for Consumer Safety, the Expert Panel for Cosmetic Ingredient Safety says it will review SCCS’s findings before issuing its own assessment.
For the second time in three years, the Personal Care Products Council is suing the California attorney general over the listing of a personal care ingredient under Proposition 65, citing a First Amendment violation.
A federal district court in Oregon denied the state’s motion to dismiss certain constitutional claims brought forward by a trade association in a suit filed last year over the state's Extended Producer Responsibility law. The case likely has implications for EPR laws across the country.
The US FDA has updated its Cosmetics Direct electronic submission portal, just as MoCRA stakeholders revisit the portal to renew facility registrations.
IRI-Sys’s AI-native software helps brands reduce the time involved in developing products so they can bring beauty products to market faster, while helping manufacturers enhance efficiency, ensure quality and scale manufacturing capabilities.
Comments on the US Food and Drug Administration’s bemotrizinol proposed order were unanimously in favor of the measure, with some stakeholders taking the opportunity to encourage the agency to adopt additional sunscreen regulatory measures to speed up approval of new filters.
The Estee Lauder Companies alleges retail giant Walmart has been ‘knowingly’ selling counterfeit versions of products from six of its brands. It filed a lawsuit against the firm Feb. 9.
A proposed hazard-based regulatory framework may complicate the opportunity for Europe’s cosmetics industry to show that cosmetic ingredients and products are safe.
The Estee Lauder Companies, Inc. pleaded guilty to two counts of violating a Canadian Act that had required it to correct missteps related to selling products containing PFAS.
“Dietary Supplement Regulatory Uniformity Act” would prohibit states from adding requirements and rules for supplement manufacturing and sales on top of FDA regulations.
The European Commission’s recent environmental omnibus failed to address industry concerns on wastewater cost allocations, and legal pathways are expected to be the way forward.
The US FDA published a Q&A draft guidance for cosmetic manufacturers on its expanded record-gathering authority under MoCRA, the types of documents that do not apply and the repercussions for stakeholders that do not comply with requests for data.
The US FDA’s recent warning letters demonstrates increased scrutiny of contract manufacturers in the OTC and cosmetics space over potential benzene formation. Separately, NGO Weaving Voices for Health & Justice continues to pressure FDA after it misses its deadline for a formaldehyde proposed rule.
Venable LLP attorney Claudia Lewis shares how MoCRA stakeholders can audit safety substantiation records, take steps toward GMP compliance and review labeling claims, while also preparing internal processes to enable a rapid response in the case of an FDA mandatory recall.
Private Label Skin Care Inc. has violated Current Good Manufacturing Practice regulations for pharmaceuticals, according to the agency.