Policy & Regulation
FDA aims to prevent poor quality or incomplete responses to Form 483 inspection observations with a new draft guidance describing the structure and content for concise, factual and effective corrective action responses.
In Part 2 of HBW Insight’s Over the Counter podcast with David Pineda Ereño, we discuss how consumer health companies are navigating fragmented EU rules and leveraging e‑commerce to exploit the commercial potential of postbiotics. We also briefly discuss the exciting area of postbiotics.
US Trade Representative is taking two swings under Section 301(b) of the 1974 Trade Act. Customs and Border Protection has March 19 to provide US Court of International Trade with next update on its progress on providing refunds for IEEPA tariffs.
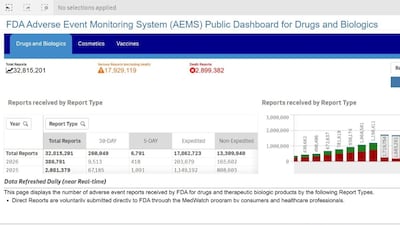
FDA launches first phase of unified platform for adverse event reports, replacing separate databases for different product types. Cosmetics, OTC drugs in initial launch; supplements start in May.
FTC’s sending checks totaling more than $40,700 to 578 consumers who purchased deceptively marketed treatment plans from Golden Sunrise between July 2017 and July 2020. Company owner and chief medical officer both pleaded guilty to criminal charges.
US Court of International Trade expects by March 12 to receive from Customs and Border Protection a “short report describing the progress” it “has made toward the development of a process to issue refunds of IEEPA duties paid with interest.”
Delaying or altogether eliminating limits on all THCs in lawful hemp Congress approved in 2025 could come down to either House Appropriations AG/FDA subcommittee, chaired by a proponent for lower limits, or Energy and Commerce Health subcommittee, chaired by a supporter for eliminating lower limits.
The second panel of this week's Geneva Self‑Care Forum explored further the tension between self‑care as a public‑health tool and self‑care as an out‑of‑pocket consumer market, raising sharp questions about where the industry sits.
Industry groups acknowledge a March 27 public meeting has potential to influence how firms can expand product lines with additional ingredients and formulations allowed under DSHEA. FDA acknowledges industry’s interest in introducing additional ingredients.
When House Agriculture Committee on March 3 considers five-farm bill year reauthorization, it will vote whether to add an amendment to extend by a year the Nov. 12 deadline for compliance with lower limits on all THC levels allowed in lawful hemp.
Bills from lawmakers in California (AB 2030) and Connecticut (SB 227) follow similar legislation being considered during current legislative sessions in Alaska, Hawaii, Massachusetts, Michigan and Washington.
Despite 2025 volume stagnation in France and Italy, structural opportunities in category penetration, wellness premiumization, pharmacy expansion, and Rx-to-OTC switch suggest the self-care market still has clear levers for growth.
GRAS observations included a report on modernizing FDA published as consumer health industry and food safety advocacy and research groups meet with FDA and OMB officials to offer recommendations for and against a proposed rule agency intends to publish on eliminating self-affirmed GRAS option.
Warning letter, submitted three months after FDA rejected supplement industry groups’ arguments for leeway around how INDs affect dietary ingredient’s availability for use in supplements, stated Prodrome Sciences’ clinical trials with omega-3 supplements should have been registered as IND.
Nearly one in five UK adults have used CBD in the past year, mainly for wellbeing, mental health and pain, with many users reporting reliance, strong brand‑trust considerations and growing concerns about unauthorized products.
After three years, EFSA reopens the CBD novel food process with a very conservative provisional safe intake of just 2mg/day due to major unresolved safety data gaps.
Agency exercises enforcement discretion on requirement for products to be labeled as containing artificial colors or dyes when those aren’t native to foods or ingredients, including added colors made from natural sources.
“Dietary Supplement Regulatory Uniformity Act” would prohibit states from adding requirements and rules for supplement manufacturing and sales on top of FDA regulations.
The latest round of updates to the European Commission's Novel Food Catalogue determine whether authorization is required for supplements containing certain botanical extracts.
Hawaii proposes age-restricted sales but, unlike similar bills also filed in Alaska, Massachusetts, Michigan and Washington legislatures, would require behind-the-counter storage to limit consumer access in stores.